- To implement more efficient and biologically-supported personalized medicine for cognitive and social alterations relevant to psychiatric and neurodevelopmental disorders.
- To develop a bidirectional mouse-human approach to accelerate the development of precision medicine strategies, with a deeper understanding of the biological mechanisms involved.
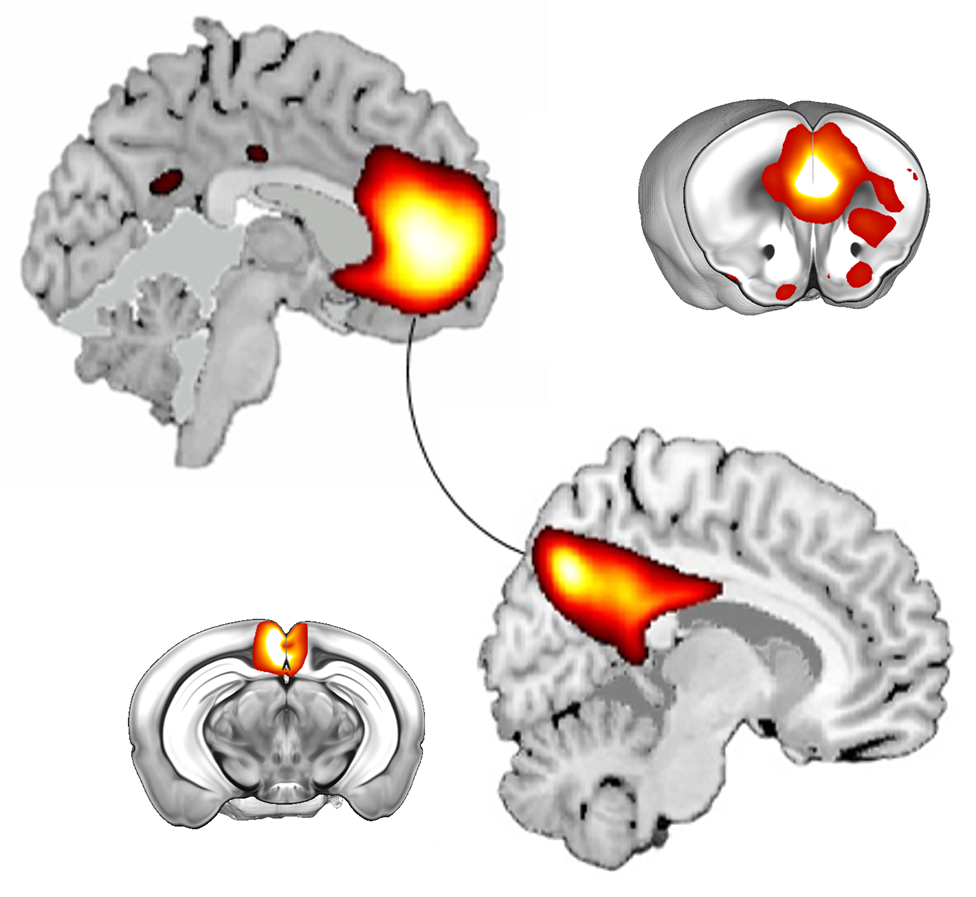
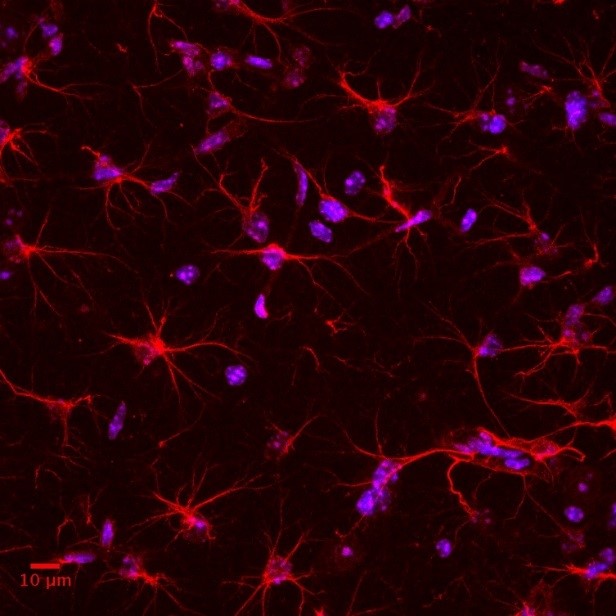
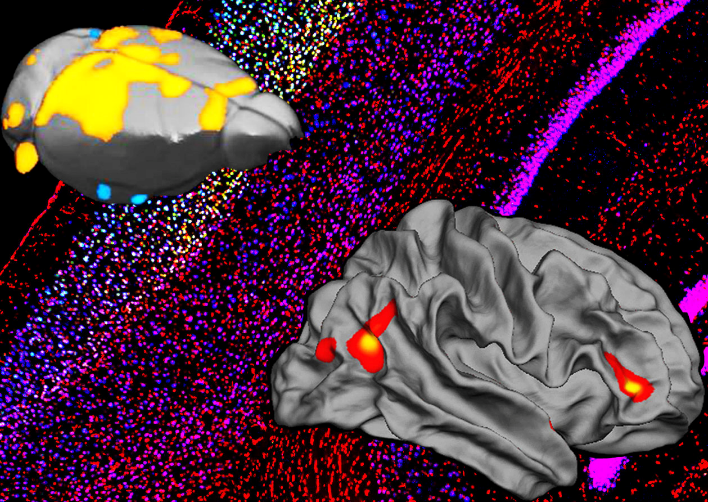
- To investigate genetics, circuits, and cell-type specific mechanisms underlying higher-order cognitive and social processes.
- To understand the brain-immune system interplay in modulating cognitive and social functions, their developmental trajectories, and the impact of early treatments.
Genetics of Cognition
Our aim is to uncover the mechanisms underlying cognitive and social alterations relevant to psychiatric and neurodevelopmental disorders with the final goal of developing biologically-based and personalized therapeutic strategies. We employ cross-disciplinary approaches including detailed in vivo studies in genetically modified mice strictly combined with clinical investigations.
Overreaching Research Goals
News
Top publications
2024
Dautan D., Monai A., Maltese F., Chang X., Molent C., Mauro D., Galbusera A., Vecchia D., Antonelli F., Benedetti A., Drago F., Leggio G.M., Pagani M., Fellin T., Gozzi A., Schumann G., Manago F., Papaleo F.
Cortico-cortical transfer of socially derived information gates emotion recognition
Nature Neuroscience
2022
Scheggia D., La Greca F., Maltese F., Chiacchierini G., Italia M., Molent C., Bernardi F., Coccia G., Carrano N., Zianni E., Gardoni F., Di Luca M., Papaleo F.
Reciprocal cortico-amygdala connections regulate prosocial and selfish choices in mice
Nature Neuroscience, vol. 25, (no. 11), pp. 1505-1518
2020
Scheggia D., Manago F., Maltese F., Bruni S., Nigro M., Dautan D., Latuske P., Contarini G., Gomez-Gonzalo M., Requie L.M., Ferretti V., Castellani G., Mauro D., Bonavia A., Carmignoto G., Yizhar O., Papaleo F.
Somatostatin interneurons in the prefrontal cortex control affective state discrimination in mice
Nature Neuroscience, vol. 23, (no. 1), pp. 47-60
2019
Ferretti V., Maltese F., Contarini G., Nigro M., Bonavia A., Huang H., Gigliucci V., Morelli G., Scheggia D., Manago F., Castellani G., Lefevre A., Cancedda L., Chini B., Grinevich V., Papaleo F.
Oxytocin Signaling in the Central Amygdala Modulates Emotion Discrimination in Mice
Current Biology, vol. 29, (no. 12), pp. 1938-1953.e6
2018
Scheggia D., Mastrogiacomo R., Mereu M., Sannino S., Straub R.E., Armando M., Manago F., Guadagna S., Piras F., Zhang F., Kleinman J.E., Hyde T.M., Kaalund S.S., Pontillo M., Orso G., Caltagirone C., Borrelli E., De Luca M.A., Vicari S., Weinberger D.R., Spalletta G., Papaleo F.
Variations in Dysbindin-1 are associated with cognitive response to antipsychotic drug treatment
Nature Communications, vol. 9, (no. 1)
Principal Investigator
Genetics of Cognition
IIT OpenTalk Magazine - Lab Highlights Show all >